
Fig. 7. Phoretic deutonymphs of Roubikia imberba on the dorsal mesosoma and metasoma of bee Tetrapedia sp. from Argentina.
Fig. 7. Phoretic deutonymphs of Roubikia imberba on the dorsal mesosoma and metasoma of bee Tetrapedia sp. from Argentina.

Fig. 8. Phoretic deutonymphs of Roubikia panamensis on the dorsal metasoma (many) and mesosoma (few) of bee Coelioxoides waltheriae.
Fig. 8. Phoretic deutonymphs of Roubikia panamensis on the dorsal metasoma (many) and mesosoma (few) of bee Coelioxoides waltheriae.
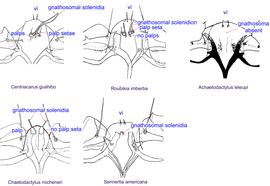
Fig. 9. Gnathosomal morphology is a key character separating genera of the family Chaetodactylidae (Centriacarus, Roubikia, Achaetodactylus, Chaetodactylus, and Sennertia) based on phoretic deutonymphs. In particular, Roubikia can be distinguished by its gnathosomal solenidion and palp setae present and free palps absent; drawing of Achaetodactylus leleupi courtesy of Belgian GTI Focal Point 2009, http://www.taxonomy.be.
Fig. 9. Gnathosomal morphology is a key character separating genera of the family Chaetodactylidae (Centriacarus, Roubikia, Achaetodactylus, Chaetodactylus, and Sennertia) based on phoretic deutonymphs. In particular, Roubikia can be distinguished by its gnathosomal solenidion and palp setae present and free palps absent; drawing of Achaetodactylus leleupi courtesy of Belgian GTI Focal Point 2009, http://www.taxonomy.be.
Fig. 10. Ventral view of phoretic deutonymph of Roubikia imberba showing closed coxal fields IV.
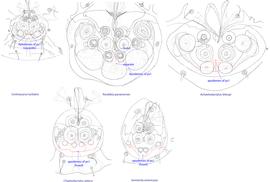
Fig. 11. Attachment organs of phoretic deutonymphs of the family Chaetodactylidae, showing the structure of apodemes of ps1. Roubikia is unique in having these apodemes fused anteriorly and separate posteriorly.
Fig. 11. Attachment organs of phoretic deutonymphs of the family Chaetodactylidae, showing the structure of apodemes of ps1. Roubikia is unique in having these apodemes fused anteriorly and separate posteriorly.
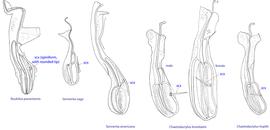
Fig. 12. Supracoxal sclerite of homeomorphic males of the family Chaetodactylidae (female supracoxal sclerite of Ch. krombeini given for comparison), showing the shape of supracoxal setae (scx). Roubikia is unique in having a spiniform scx with rounded tip, while in all other genera scx is filiform with a pointed tip.
Fig. 12. Supracoxal sclerite of homeomorphic males of the family Chaetodactylidae (female supracoxal sclerite of Ch. krombeini given for comparison), showing the shape of supracoxal setae (scx). Roubikia is unique in having a spiniform scx with rounded tip, while in all other genera scx is filiform with a pointed tip.
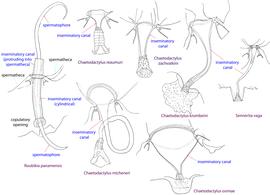
Fig. 13. Spermatheca and inseminatory canal of Chaetodactylidae. Roubikia is unique in having a cylindrical inseminatory canal that protrudes into the spermatheca and distinct, filariform spermatophores. In other chaetodactylid genera, the inseminatory canal is distinctly widened at its entrance to the spermatheca and does not protrude into the spermatheca; spermatophores are absent.
Fig. 13. Spermatheca and inseminatory canal of Chaetodactylidae. Roubikia is unique in having a cylindrical inseminatory canal that protrudes into the spermatheca and distinct, filariform spermatophores. In other chaetodactylid genera, the inseminatory canal is distinctly widened at its entrance to the spermatheca and does not protrude into the spermatheca; spermatophores are absent.
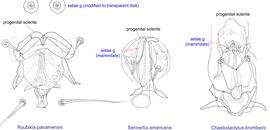
Fig. 14. Male genital apparatus and genital setae (g) of Chaetodactylidae. Roubikia is unique in having genital setae modified to transparent disks, while in other genera these setae are mammillate.
Fig. 14. Male genital apparatus and genital setae (g) of Chaetodactylidae. Roubikia is unique in having genital setae modified to transparent disks, while in other genera these setae are mammillate.
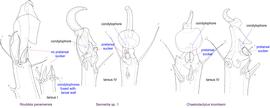
Fig. 15. Leg tarsi of males of Chaetodactylidae. Roubikia can be distinguished from other chaetodactylid genera with known males (Sennertia and Chaetodactylus) by the absence of pretarsal suckers. In the other two genera, male pretarsal suckers are present. For Roubikia heteromorphic males are shown, for the others homeomorphic males.
Fig. 15. Leg tarsi of males of Chaetodactylidae. Roubikia can be distinguished from other chaetodactylid genera with known males (Sennertia and Chaetodactylus) by the absence of pretarsal suckers. In the other two genera, male pretarsal suckers are present. For Roubikia heteromorphic males are shown, for the others homeomorphic males.
Fig. 5. Sancassania boharti phoretic deutonymph legs III-IV, lateral view, and attachment organ.
Fig. 15. Phoretic deutonymphs of Sancassania boharti on male alkali bee Nomia melanderi.

Fig. 16. Phoretic deutonymphs of Sancassania boharti in the intersegmental space of sternite 1 and tergite 1 (S1 and T1) of male bee Nomia melanderi.
Fig. 16. Phoretic deutonymphs of Sancassania boharti in the intersegmental space of sternite 1 and tergite 1 (S1 and T1) of male bee Nomia melanderi.

Fig. 17. Sancassania sp. adults and eggs. Culture established from deutonymphs collected from Pelidnota punctata.
Fig. 17. Sancassania sp. adults and eggs. Culture established from deutonymphs collected from Pelidnota punctata.

Fig. 18. Sancassania sp. feeding stages. Culture established from deutonymphs collected from Pelidnota punctata.
Fig. 18. Sancassania sp. feeding stages. Culture established from deutonymphs collected from Pelidnota punctata.
Fig. 3. Saproglyphus sp. (ex Osmia aquilonaria) phoretic deutonymph idiosoma, ventral view.